 
Cycles or episodes of VBD disease incidence are driven by a system of interconnected vector, host, and pathogen population abundances that vary over time and space. Vector-borne diseases (VBDs) remain a serious threat to human health ( San Martín et al., 2010 Dick et al., 2012 Lee et al., 2013 Mead, 2015 CDC, 2016 Faria et al., 2016), livestock ( Wilson and Mellor, 2009), and agriculture ( Taylor et al., 2016). 
Determining when and to what extent it is important to incorporate trait variation into vector borne disease models remains an important, outstanding question. This framework provides a means to conceptualize how traits can be incorporated in vector borne disease systems, and identifies key areas in which trait variation can be explored. We provide a conceptual framework for incorporating trait variation into vector-borne disease transmission models, and highlight key empirical and theoretical challenges. We argue that mechanistically incorporating trait variation into these models, by explicitly capturing its effects on vector fitness and abundance, can improve the reliability of their predictions in a changing world. Here, we review empirical evidence for variation in vector traits and how this trait variation is currently incorporated into mathematical models of vector-borne disease transmission. Increasing empirical evidence shows that vector traits vary significantly across individuals, populations, and environmental conditions, and at time scales relevant to disease transmission dynamics. 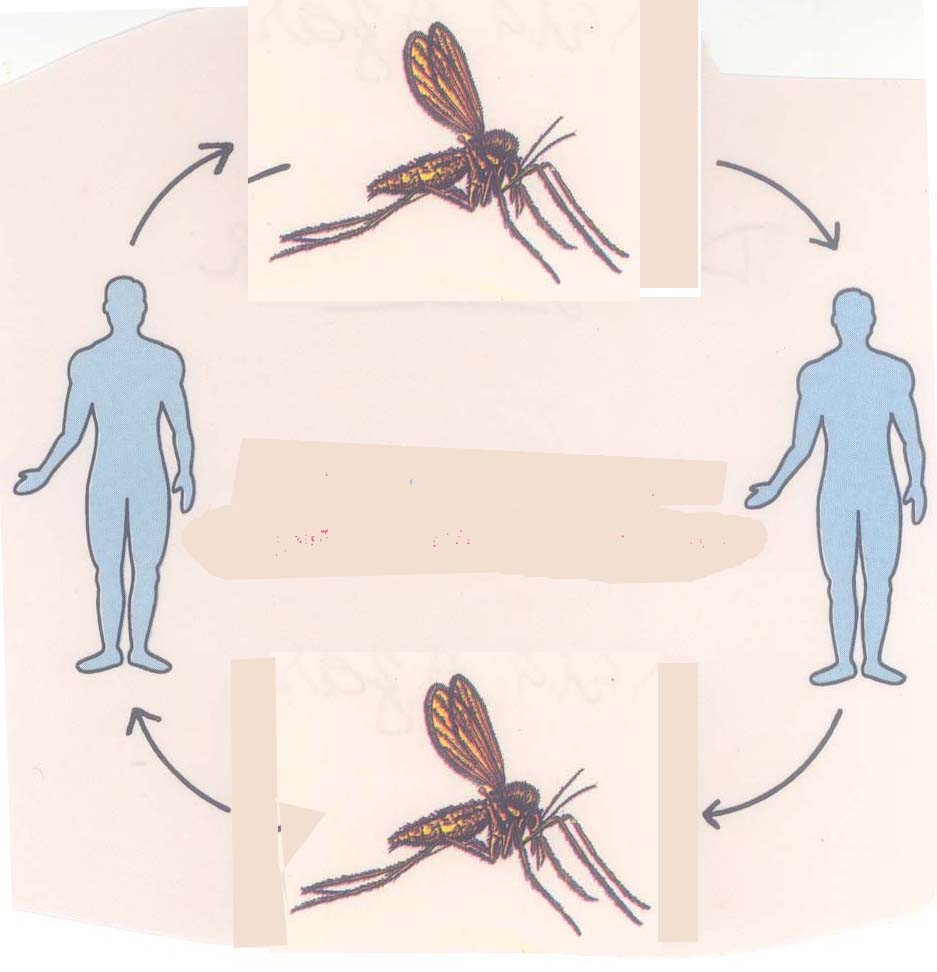
The functional traits of vectors can affect pathogen transmission rates directly and also through their effect on vector population dynamics. Many important endemic and emerging diseases are transmitted by vectors that are biting arthropods.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
